Vero Cell Vaccine Covid-19 Effectiveness Percentage, Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
The addition of this vaccine has the potential to rapidly accelerate COVID-19 vaccine access for countries seeking to. Results of a prospective randomized trial Vaccine.
World Health Organization Who What S The Difference Between Covid 19 Vaccine Efficacy And Effectiveness Vaccine Efficacy Refers To How The Vaccine Performs In Ideal Conditions Controlled Clinical Trials Vaccine
Vero Cell is produced by Beijing Bio-Institute of Biological Products Co Ltd a subsidiary of China National Biotec Group CNBG.
Vero cell vaccine covid-19 effectiveness percentage. BBIBP-CorV also known as the Sinopharm COVID-19 vaccine or. However this vaccine is an inactivated vaccine with an adjuvant that is commonly used in many other vaccines with a well-documented safety profile such as Hepatitis B and Tetanus vaccines including. While mRNA vaccines such as the PfizerBioNTech COVID-19 vaccine and Moderna COVID-19 vaccine showed higher efficacy of 90 mRNA vaccines present distribution challenges for some nations as some may require deep-freeze facilities and trucks.
Providing Covid-19 vaccines to outgoing migrants was necessary. Stratified precipitate may form which. This is operationalized as a reduction in hospitalization and death.
Listing a study does not mean it has been evaluated by the US. Providing the Vero Cell vaccine to outbound migrants. The manufacturer has not released official figures on the efficacy of the CoronaVac vaccine but evaluations by the Brazilian collaborators suggest an efficacy of up to 78.
An interim analysis reveals the Covaxin BBV152 vaccine was relatively safe and 81 percent effective in preventing symptomatic COVID-19. COVID-19 Vaccine Vero Cell Inactivated COMPOSITION Active ingredient. New analysis by PHE shows for the first time that 2 doses of COVID-19 vaccines are highly effective against hospitalisation from the Delta B16172 variant.
Adjuvanted inactivated vaccine produced in Vero cells and developed by India showed 81 efficacy in preventing COVID-19 after two doses. Estimating vaccine efficacy for COVID-19 projections. Listing a study does not mean it has been evaluated by the US.
Purified Vero cell rabies vaccine is safe carries a very low adverse reaction rate and is effective in preventing rabies in severely exposed subjects when used with human or equine rabies immune globulin. The World Health Organization WHO has listed Sinopharms Vero Cell COVID-19 vaccine for emergency use allowing it to be rolled out globally. Efficacy Safety and Immunogenicity of Inactivated SARS-CoV-2 Vaccines Vero Cell to Prevent COVID-19 in Healthy Adult Population In Peru Healthy Adult Population In Peru Covid-Peru The safety and scientific validity of this study is the responsibility of the study sponsor and investigators.
Efficacy at preventing infection. Doses of the Chinese Sinopharm vaccine against the coronavirus disease COVID-19 are seen at the Biblioteka kod Milutina restaurant in Kragujevac Serbia May 4 2021. This is operationalized as a reduction in susceptibility.
The CHMPs decision to start the rolling review is based on preliminary results from laboratory studies non-clinical data and clinical studies. COVID-19 Vaccine Vero Cell Inactivated Sinopharm COVID-19 Vaccine Explainer 24 MAY 2021 Special population groups based on available data as of 7 May 2021 For persons with comorbidities phase 3 clinical trial data are insufficient to determine vaccine efficacy. The efficacy rating followed an interim report of ongoing human trials conducted in that country CNBC reported.
Disodium hydrogen phosphate dodecahydra te sodium dihydrogen phosphate monohydrate sodium chloride DESCRIPTION CoronaVac is a milky-white suspension. Know the risks and potential. Inferring from other inactivated vaccines.
Efficacy at preventing symptomatic disease. Its product name is SARS-CoV-2 Vaccine Vero Cell not. EMAs human medicines committee has started a rolling review of COVID-19 Vaccine Vero Cell Inactivated developed by Sinovac Life Sciences Co LtdThe EU applicant for this medicine is LifeOn Srl.
A Study to Evaluate The Efficacy Safety and Immunogenicity of Inactivated SARS-CoV-2 Vaccines Vero Cell in Healthy Population Aged 18 Years Old and Above COVID-19 The safety and scientific validity of this study is the responsibility of the study sponsor and investigators. Currently the IHME model uses the following inputs of vaccine efficacy separated by variant. The available data on the Sinovac-CoronaVac COVID-19 vaccine in pregnant women are insufficient to assess either vaccine efficacy or possible vaccine-associated risks in pregnancy.
But now there is a new problem said Sujit Kumar Shrestha general secretary of Nepal Association of Foreign Employment Agencies NAFEA an umbrella organisation of privately-run recruiting agencies hiring and supplying Nepali migrant workers to foreign employers. At present our model only distinguishes. Dec 09 2020 By Alex Keown The COVID-19 vaccine under development by Chinas Sinopharm is showing efficacy of 86 health authorities from the United Arab Emirates reported this morning.
Inactivated SARS-CoV-2 Virus CZ02 s train Adjuvant. Safety and efficacy of purified Vero cell rabies vaccine given intramuscularly and intradermally. A large register-based study on prevention of COVID-19 transmission in households of vaccinated healthcare workers from Scotland suggests that vaccination of a household member reduces the risk of infection in susceptible household members by at least 30.
Estimated effectiveness of Moderna COVID-19 vaccine Studies mostly estimate a similar protection level as the Pfizer-BioNTech vaccine with numbers ranging from 66 percent to 95 percent.

Interim Estimates Of Vaccine Effectiveness Of Bnt162b2 And Mrna 1273 Covid 19 Vaccines In Preventing Sars Cov 2 Infection Among Health Care Personnel First Responders And Other Essential And Frontline Workers Eight U S Locations December
Who Lists Sinopharm S Covid 19 Vaccine For Emergency Use
Coronavirus Covaxin Efficacy Is 81 Works Against Variants The Hindu

Efficacy Of Sinopharm S Covid 19 Vaccines Proved Again In New Trials Cgtn

Sinopharm Vero Cell Inactivated Covid 19 Vaccine
Chinese Pharmaceutical Firm Sinovac Says Its Covid 19 Vaccine Is More Than 50 Percent Effective But Data Withheld The Boston Globe
China S Covid Vaccine From Sinopharm Is 86 Effective Uae Says
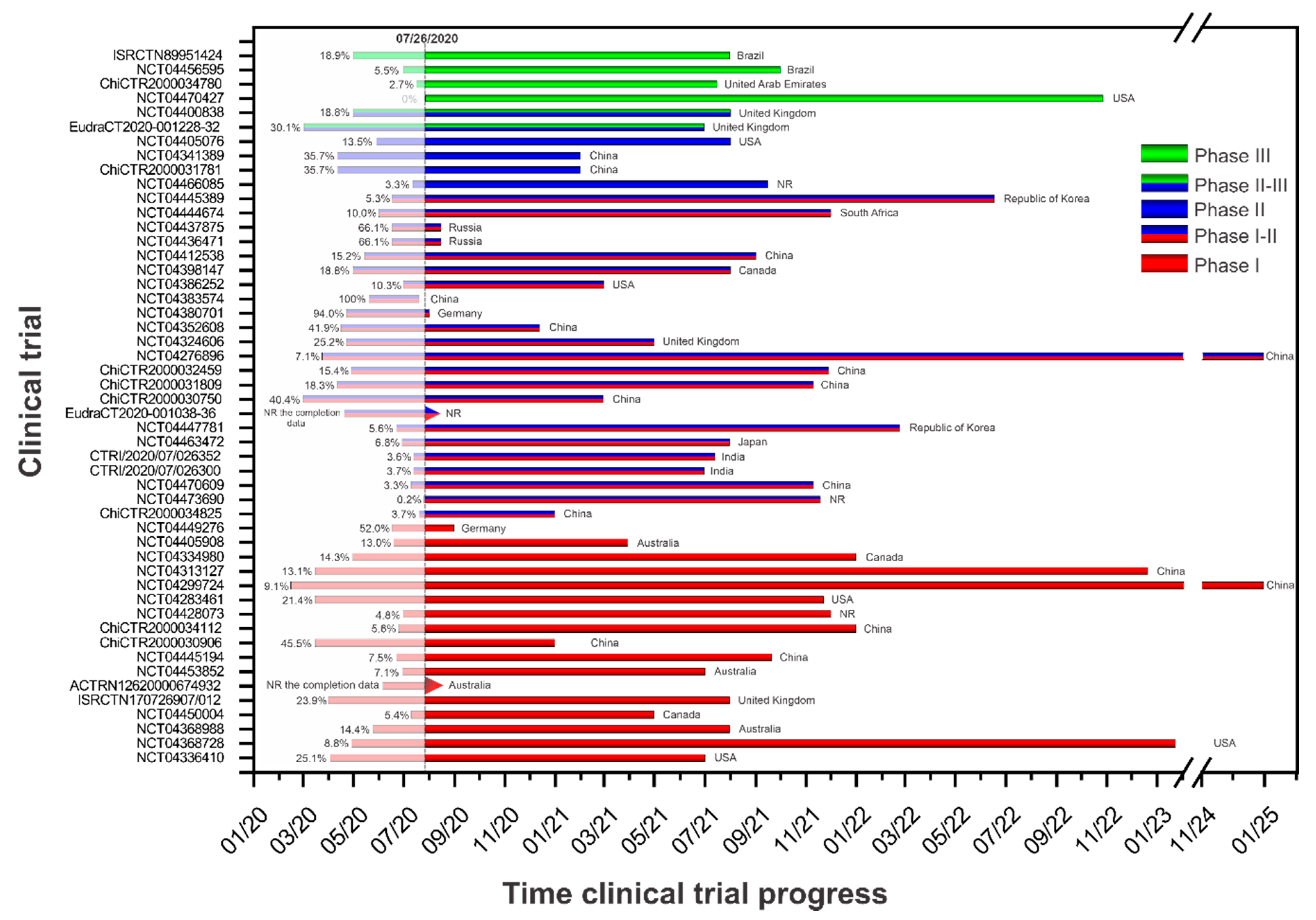
Vaccines Free Full Text Current Clinical Trials Protocols And The Global Effort For Immunization Against Sars Cov 2 Html
Sinopharm S Two Covid 19 Shots Effective Study Says Reuters
Singapore Excludes Sinovac Shots From Covid 19 Vaccination Tally Nikkei Asia

Efficacy And Safety Of An Inactivated Whole Virion Sars Cov 2 Vaccine Coronavac Interim Results Of A Double Blind Randomised Placebo Controlled Phase 3 Trial In Turkey The Lancet
Who Approves China S Sinopharm Covid 19 Vaccine For Emergency Use Has 79 Efficacy Coronavirus Outbreak News
Sinopharm S Covid 19 Vaccine Shows 86 Efficacy Uae Health Agency Says Biospace
Https Cdn Who Int Media Docs Default Source Immunization Sage 2021 April 1 Sage29apr2021 Sinopharm Pdf Sfvrsn Ddf0d841 5
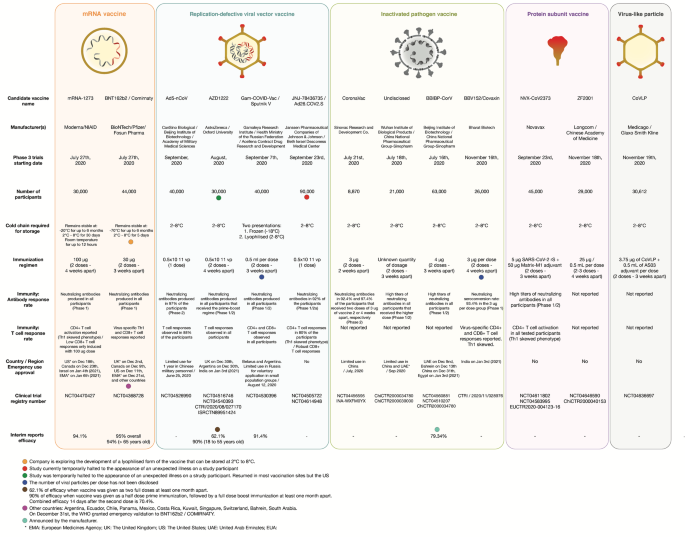
Sars Cov 2 Vaccines Strategies A Comprehensive Review Of Phase 3 Candidates Npj Vaccines
Peru Study Finds Sinopharm Covid Vaccine 50 4 Effective Against Infections Dhaka Tribune
Https Cdn Who Int Media Docs Default Source Immunization Sage 2021 April 1 Sage29apr2021 Sinopharm Pdf Sfvrsn Ddf0d841 5
Uae Says Sinopharm Vaccine Has 86 Efficacy Against Covid 19 Reuters
Coronavirus Who Approves Sinovac Covid Vaccine For Emergency Use News Dw 01 06 2021


.JPG)

Post a Comment
Post a Comment